 The greater the masses of attached atoms, the lower the IR frequency at which the bond will absorb. To be more precise, it is the masses of the two atoms which are of greater importance. 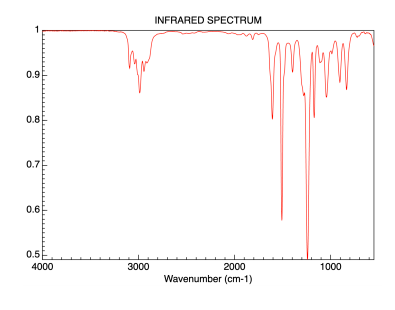
The following topics cover some of the most important of these principles.Īs mentioned previously, one of the major factors influencing the IR absorption frequency of a bond are the identity of the two atoms involved. This complex lower region is also known as the "fingerprint region" because almost every organic compound produces a unique pattern in this area - Therefore identity can often be confirmed by comparison of this region to a known spectrum.Īlthough the above and similar IR absorption tables provide a good starting point for assigning simple IR spectra, it is often necessary to understand in greater detail some more specific properties of IR spectra. Two signals which can be seen clearly in this area is the carbonyl group, which is a very strong peak around 1700 cm -1, and the C-O bond with can be one or two strong peaks around 1200 cm -1. In contrast, the right half of the spectrum, below 2000 cm -1, normally contains many peaks of varying intensities, many of which are not readily identifiable. The frequencies from 2800 to 2000 cm -1 are normally void of other absorptions, so the presence of alkyne or nitrile groups can be easily seen here. A very broad peak in the region between 31 cm -1 indicates the presence of exchangeable protons, typically from alcohol, amine, amide or carboxylic acid groups (see further discussion of this below). First, alkane C-H stretching absorptions just below 3000 cm -1 demonstrate the presence of saturated carbons, and signals just above 3000 cm -1 demonstrate unsaturation. The left half, above 2000 cm -1, usually contains relatively few peaks, but some very diagnostic information can be found here. Upon first inspection, a typical infrared spectrum can be visually divided into two regions. These trends in aborption can be further summarized into the following categories Occasionally absorption frequency is given as a single approximation denoted with an ~ rather than a range. A broad signal shape is sometimes indicated by br.  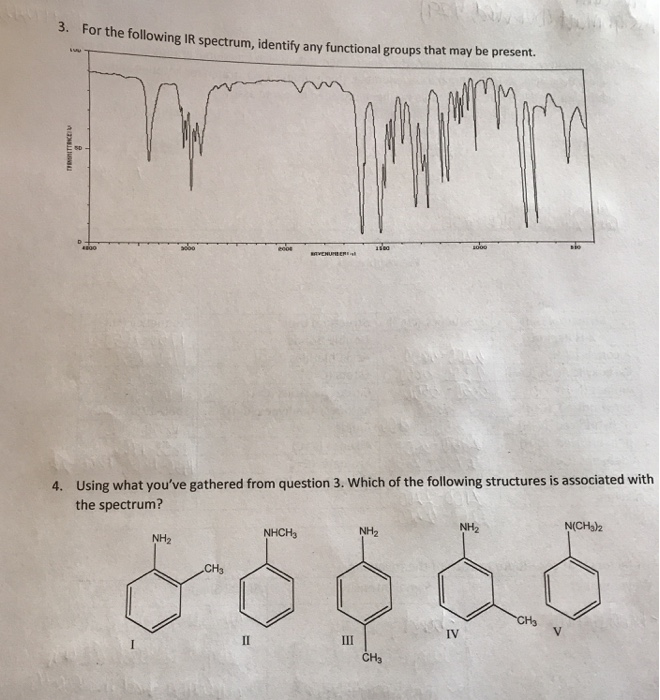
In IR absorption tables, signal intensities (height) are usually denoted by the following abbreviations: w = weak, m = medium, s = strong, v = variable. Consequently tables of IR absorptions are arranged by functional group - it some versions these may be further subdivided to give more precise information. Therefore the same or similar functional groups in different molecules will typically absorb within the same, specific frequency ranges. Conjugation and nearby atoms shift the frequency to a lesser degree. As a general rule, the most important factors determining where a chemical bond will absorb are the bond order and the types of atoms joined by the bond. These data can be quickly referenced through tables of IR absorption ranges and compared to the spectrum under consideration. Over time organic chemists have recorded and catalogued the types and locations of IR absorptions produced by a wide variety of chemical bonds in various chemical environments. For example, neither of the carbon-carbon bonds in ethene or ethyne absorb IR radiation. It is also important to note that symmetrical vibrations do not cause absorption of IR radiation. Stretching absorptions usually produce stronger peaks than bending, however the weaker bending absorptions can be useful in differentiating similar types of bonds (e.g. Thus IR spectroscopy involves collecting absorption information and analyzing it in the form of a spectrum - The frequencies at which there are absorptions of IR radiation ("peaks" or "signals") can be correlated directly to bonds within the compound in question.īecause each interatomic bond may vibrate in several different motions (stretching or bending), individual bonds may absorb at more than one IR frequency. Chemical bonds in different environments will absorb varying intensities and at varying frequencies. Radiation in this region can be utilized in organic structure determination by making use of the fact that it is absorbed by interatomic bonds in organic compounds. 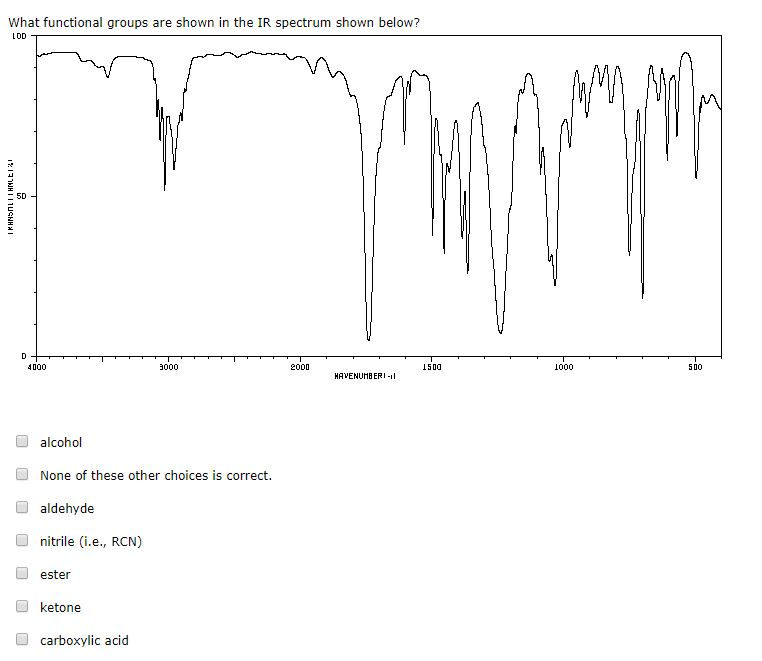
The category of EM radiation is termed infrared (IR) radiation, and its application to organic chemistry known as IR spectroscopy. IR SPECTRUM FUNCTIONAL GROUPS VERIFICATIONIntroduction to IR Spectra Introduction to IR SpectraĪn invaluable tool in organic structure determination and verification involves the class of electromagnetic (EM) radiation with frequencies between 4000 and 400 cm -1 (wavenumbers). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
